References
1. Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988;31:315-24.
2. Nienhuis RLF, Mandema E. A new serum factor in patients with rheumatoid arthritis: the perinuclear factor. Ann Rheum Dis 1964;23:302-5.
3. van Boekel MAM, Vossenaar ER, van den Hoogen FHJ, van Venrooij WJ. Auto-antibody systems in rheumatoid arthritis: specificity, sensitivity and diagnostic value. Arthritis Res 2002;4:87-93.
4. Young BJJ, Mallya RK, Leslie RDG, Clark CJM, Hamblin TJ. Anti-keratin antibodies in rheumatoid arthritis. Br Med J 1979;2:97-9.
5. Simon M, Girbal E, Sebbag M, Gomes-Daudrix V, Vincent C, Salam G, Serre G. The cytokeratin filament-aggregating protein filaggrin is the target of the so-called „antikeratin antibodies“, autoantibodies specific for rheumatoid arthritis. J Clin Invest 1993;92:1387-93.
6. Sebbag M, Simon M, Vincent C, Masson-Bessiere C, Girbal E, Durieux JJ, Serre G. The antiperinuclear factor and the so-called antikeratin antibodies are the same rheumatoid arthritis-specific autoantibodies. J Clin Invest 1995;95:2672-9.
7. Schellekens GA, de Jong BAW, van den Hoogen FHJ, van de Putte LBA, van Venrooij WJ. Citrulline is an essential constituent of antigenic determinants recognized by rheumatoid arthritis-specific autoantibodies. J Clin Invest 1998;101:273-81.
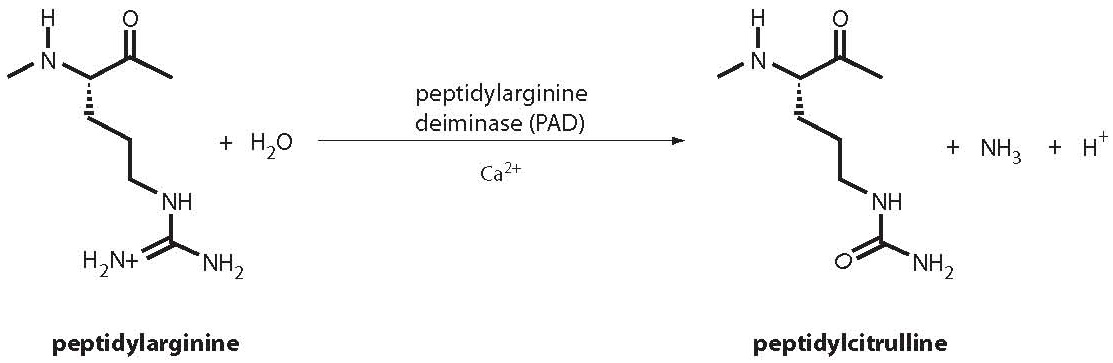
8. Girbal-Neuhauser E, Durieux JJ, Arnaud M, Dalbon P, Sebbag M, Vincent C, et al. The epitopes targeted by the rheumatoid arthritis-associated antifilaggrin autoantibodies are posttranslationally generated on various sites of (pro)filaggrin by deimination of arginine residues. J Immunol 1999;162:585-94.
9. Cantaert T, De Rycke L, Bongartz T, Matteson EL, Tak PP, Nicholas AP, Baeten D. Citrullinated proteins in rheumatoid arthritis. Arthritis Rheum 2006;54:3381-9.
10. Masson-Bessiere C, Sebbag M, Durieux JJ, Nogueira L, Vincent C, Girbal-Neuhauser E, et al. In the rheumatoid pannus, anti-filaggrin autoantibodies are produced by local plasma cells and constitute a higher proportion of IgG than in synovial fluid and serum. Clin Exp Immunol 2000;119:544-52.
11. Masson-Bessiere C, Sebbag M, Girbal-Neuhauser E, Nogueira L, Vincent C, Senshu T, Serre G. The major synovial targets of the rheumatoid arthritis-specific antifilaggrin autoantibodies are deiminated forms of the alpha- and beta-chains of fibrin. J Immunol 2001;166:4177-84.
12. Chapuy-Regaud S, Sebbag M, Baeten D, Clavel C, Foulquier C, De Keyser F, Serre G. Fibrin deimination in synovial tissue is not specific for rheumatoid arthritis but commonly occurs during synovitides. J Immunol 2005;174:5057-64.
13. Suzuki A, Yamad R, Ohtake-Yamanaka M, Okazaki Y, Sawada T, Yamamoto K. Anti-citrullinated collagen type I antibody is a target of autoimmunity in rheumatoid arthritis. Biochem Biophys Res Commun 2005;333:418-26.
14. Vossenaar ER, Despres N, Lapointe E, van der Heijden A, Lora M, Senshu T, et al. Rheumatoid arthritis specific anti-Sa antibodies target citrullinated vimentin. Arthritis Res Ther 2004;6:R142-50.
15. Chang X, Yamada R, Suzuki A, Kochi Y, Sawada T, Yamamoto K. Citrullination of fibronectin in rheumatoid arthritis synovial tissue. Rheumatology 2005;44:1374-82.
16. Kinloch A, Tatzer V, Wait R, Peston D, Lundberg K, Donatien P, et al. Identification of citrullinated α-enolase as a candidate autoantigen in rheumatoid arthritis. Arthritis Res Ther 2005;7:R1421-9.
17. Pratesi F, Tommasi C, Anzilotti C, Chimenti D, Migliorini P. Deiminated Epstein-Barr virus nuclear antigen 1 is a target of anti-citrullinated protein antibodies in rheumatoid arthritis. Arthritis Rheum 2006;54:733-41.
18. Nakashima K, Hagiwara T, Yamada M. Nuclear localization of peptidylarginine deiminase V and histone deimination in granulocytes. J Biol Chem 2002;277:49562-8.
19. Vossenaar ER, Zendman AJW, van Venrooij WJ, Pruijn G. PAD, a growing family of citrullinating enzymes: genes, features and involvement in disease. BioEssays 2003;25:1106-18.
20. Chapuy-Regaud S, Sebbag M, Nachat R, Baeten D, Foulquier V, Simon M, et al. Peptidylarginine deiminase isoforms expressed in the synovial membrane of rheumatoid arthritis patients. Arthritis Res Ther 2003;5(Suppl 1):5. Abstract from 23rd European Workshop for Rheumatology Research.
21. Nicholas AP, Whitaker JN. Preparation of a monoclonal antibody to citrullinated epitopes: its characterization and some applications to immunohistochemistry in human brain. Glia 2002;37:328-36.
22. Smeets TJ, Vossenaar ER, vanVenrooij WJ, Tak PP. Is expression of intracellular citrullinated proteins in synovial tissue specific for rheumatoid arthritis? Comment on the article by Baeten et al. Arthritis Rheum 2002;46:2824-6.
23. Ishigami A, Ohsawa T, Hiratsuka M, Taguchi H, Kobayashi S, Saito Y, et al. Abnormal accumulation of citrullinated proteins catalyzed by peptidylarginine deiminase in hippocampal extracts from patients with Alzheimer’s disease. J Neurosci Res 2005;80:120-8.
24. Nicholas AP, Sambandam T, Echols JD, Tourtellotte WW. Increased citrullinated glial fibrillary acidic protein in secondary progressive multiple sclerosis. J Comp Neurol 2004;473:128-36.
25. Makrygiannakis D, af Klint E, Lundberg IE, Lofberg R, Ulfgren AK, Klareskog L, Catrina AI. Citrullination is an inflammation-dependent process. Ann Rheum Dis 2006;65:1219-22.
26. Vossenaar ER, Zedman AJW, van Venrooij WJ. Citrullination, a possible functional link between susceptibility genes and rheumatoid arthritis. Arthritis Res Ther 2004;6:1-5.
27. Suzuki A, Yamada R, Chang X, Tokuhiro S, Sawada T, Suzuki M, et al. Functional haplotypes of PADI4, encoding citrullinating enzyme peptidylarginine deiminase 4, are associated with rheumatoid arthritis. Nat Genet 2003;34:395-402.
28. Migliorini P, Pratesi F, Tommasi C, Anzilotti C. The immune response to citrullinated antigens in autoimmune diseases. Autoimmun Rev 2005;4:561-4.
29. Hill JA, Southwood S, Sette A, Jevnikar AM, Bell DA, Cairns E. Cutting edge: the conversion of arginine to citrulline allows for a high-affinity peptide interaction with the rheumatoid arthritis-associated HLA-DRB1*0401 MHC class II molecule. J Immunol 2003;171:538-41.
30. Lard LR, van Gaalen FA, Schonkeren JJM, Pieterman EJ, Stoeken G, Vos K, et al. Association of the -2849 interleukin-10 promoter polymorphism with autoantibody production and joint destruction in rheumatoid arthritis. Arthritis Rheum 2003;47:1841-8.
31. Klareskog L, Lorentzen J, Padykov L, Alfredsson L. Genes and environment in arthritis: can RA be prevented? Arthritis Res 2002;4(Suppl 3):S31-S36.
32. De Rycke L, Nicholas A P, Cantaert T, Kruithof E, Echols J D, Vandekerckhove B, et al. Synovial intracellular citrullinated proteins colocalizing with peptidyl arginine deiminase as pathophysiologically relevant antigenic determinants of rheumatoid arthritis-specific humoral autoimmunity. Arthritis Rheum 2005;52:2323-30.
33. Slack SL, Mannik M, Dale BA. Diagnostic value of antibodies to filaggrin in rheumatoid arthritis. J Rheumatol 1998;25:847-51.
34. Palouso T, Lukka M, Alenius H, Kalkkinen N, Aho K, Kurki P, et al. Purification of filaggrin from human epidermis and measurement of antifilaggrin autoantibodies in sera from patients with rheumatoid arthritis by an enzyme-linked immunosorbent assay. Int Arch Allergy Immunol 1998;115:294-302.
35. Schellekens GA, Visser H, de Jong BA, van den Hoogen FH, Hazes JM, Breedveld FC, van Venrooij WJ. The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide.Arthritis Rheum 2000;43:155-63.
36. Avouac J, Gossec L, Dougados M. Diagnostic and predictive value of anti-cyclic citrullinated protein antibodies in rheumatoid arthritis: a systematic literature review. Ann Rheum Dis 2006;65:845-51.
37. Nijenhuis S, Zendman AJ, Vossenaar ER, Pruijn GJ, vanVenrooij WJ. Autoantibodies to citrullinated proteins in rheumatoid arthritis: clinical performance and biochemical aspects of an RA-specific marker. Clin Chim Acta 2004;350:17-34.
38. Burlingame RW. QUANTA LiteTM CCP 3.1 IgG/IgA ELISA. INOVA Newsletter 2007;2:4-5.
39. Vieira LMEA. Rheumatoid arthritis diagnosis: a comparative study of second and third generation anti-cyclic citrullinated peptide (CCP) antibody ELISAs. INOVA Newsletter 2007;2:8-9.
40. Lutteri L, Malaise M, Chapelle JP. Comparison of second- and third-generation anti-cyclic citrullinated peptide antibodies assays for detecting rheumatoid arthritis. Clin Chim Acta 2007;386:76-81.
41. Caro-Oleas JL, Fernandez-Suarez A, Cesteros SR, Porrino C, Nunez-Roldan A, Wichmann Schlipf I. Diagnostic usefulness of a third-generation anti-cyclic citrulline antibody test in patients with recent-onset polyarthritis. Clin Chem Lab Med 2007;45:1396-401.
42. Riedmann JP, Munoz S, Kavanaugh A. The use of second generation anti-CCP antibody (anti-CCP2) testing in rheumatoid arthritis – a systematic review. Clin Exp Rheumatol 2005;23(Suppl. 39):S69-76.
43. Rantapaa-Dahlqvist S, de Jong BA, Berglin E, Hallmans G, Wadell G, Stenlund H, et al. Antibodies against cyclic citrullinated peptide and IgA rheumatoid factor predict the development of rheumatoid arthritis. Arthritis Rheum 2003;48:2741-9.
44. Berglin E, Padyukov L, Sundin U, Hallmans G, Stenlund H, Van Venrooij WJ. A combination of autoantibodies to cyclic citrullinated peptide (CCP) and HLA-DRB1 locus antigens is strongly associated with future onset of rheumatoid arthritis. Arthritis Res Ther 2004;6:R303-8.
45. Van Gaalen FA, Linn-Rasker SP, van Venrooij WJ, de Jong BAW, Breedveld FC, Verweij CL, et al. Autoantibodies to cyclic citrullinated peptides predict progression to rheumatoid arthritis in patients with undifferentiated arthritis. Arthritis Rheum 2004;50:709-15.
46. Raza K, Breese M, Nightingale P, Kumar K, Potter T, Carruthers DM, et al. Predictive value of antibodies to cyclic citrullinated peptide in patients with very early inflammatory arthritis. J Rheumatol 2005;32:231-8.
47. Kastbom A, Strandberg G, Lindroos A, Skogh T. Anti-CCP antibody test predicts the disease course during 3 years in early rheumatoid arthritis (the Swedish TIRA project). Ann Rheum Dis 2004;63:1085-9.
48. Forslind K, Ahlmen M, Eberhardt K, Hafström I, Svensson B, for the BARFOT study group. Prediction of radiological outcome in early rheumatoid arthritis in clinical practice: role of antibodies to citrullinated peptides (anti-CCP). Ann Rheum Dis 2004;63:1090-5.
49. Rönnelid J, Wick MC, Lampa J, Lindblad S, Nordmark B, Klareskog L, van Vollenhoven RF. Longitudinal analysis of citrullinated protein/peptide antibodies (anti-CP) during 5 year follow up in early rheumatoid arthritis: anti-CP status predicts worse disease activity and greater radiological progression. Ann Rheum Dis 2005;64:1744-9.
50. Syversen SW, Gaarder PI, Goll GL, Odegard S, Haavardsholm EA, Mowinckel P, et al. High anti-cyclic citrullinated peptide levels and an algorithm of four variables predict radiographic progression in patients with rheumatoid arthritis: results from a 10-year longitudinal study. Ann Rheum Dis 2008;67:212-7.
51. Despres N, Boire G, Lopez-Longo FJ, Menard HA. The Sa system: a novel antigen-antibody system specific for rheumatoid arthritis. J Rheumatol 1994;21:1027-33.
52. Menard HA, Lapointe E, Rochdi MD, Zhou ZJ. Insights into rheumatoid arthritis derived from the Sa immune system. Arthritis Res 2000;2:429-32.
53. Strelkov SV, Herrmann H, Aebi U. Molecular architecture of intermediate filaments. Bioassays 2003;25:243-51.
54. Vossenar ER, Radstake TR, van der Heijden A, van Mansum MA, Dieteren C, de Rooij DJ, et al. Expression and activity of citrullinating peptidylarginine deiminase enzymes in monocytes and macrophages. Ann Rheum Dis 2004;63:373-81.
55. Dejaco C, Klotz W, Larcher H, Duftner C, Schirmer M, Herold M. Diagnostic value of antibodies against a modified citrullinated vimentin in rheumatoid arthritis. Arthritis Res Ther 2006;8:R119.
56. Soos L, Szekanecz Z, Szabo Z, Fekete A, Zeher M, Horvath IF, et al. Clinical evaluation of anti-mutated citrullinated vimentin by ELISA in rheumatoid arthritis. J Rheumatol 2007;34:1658-63.
57. Mathsson L, Mullazehi M, Wick MC, Sjöberg O, van Vollenhoven R, Klareskog L, Rönnelid J. Antibodies against citrullinated vimentin in rheumatoid arthritis. Higher sensitivity and extended prognostic value concerning future radiographic progression as compared with antibodies against cyclic citrullinated peptides. Arthritis Rheum 2008;58:36-45.
58. Innala L, Kokkonen H, Eriksson C, Jiddell E, Berglin E, Rantapaa-Dahlqvist S. Antibodies against mutated citrullinated vimentin are a better predictor of disease activity at 24 months in early rheumatoid arthritis than antibodies against cyclic citrullinated peptides. J Rheumatol 2008;35:1-7.