References
1. Welsch C, Nagasawa H. Prolactin and murine mammary tumorigenesis: a review. Cancer Res 1977;37:951-63.
2. Mershon J, Sall W, Mitchner N, Ben-Jonathan N. Prolactin is a local growth factor in rat mammary tumors. Endocrinology 1995;136: 3619-23.
3. Imagawa W, Tomooka Y, Hamamoto S, Nandi S. Stimulation of mammary epithelial cell growth in vitro: interaction of epidermal growth factor and mammogenic hormones. Endocrinology 1995;116:1514-24.
4. Vonderhaar BK. Prolactin: transport, function, ad receptors in mammary gland development and differentiation. In: Neville MC, Daniel CW, eds. The mammary gland. New York (NY): Plenum Publishing Corporation; 1987;383-483.
5. Das R, Vonderhaar BK. Prolactin as a mitogen in mammary cells. J Mammary Gland Biol Neoplasia 1997;2:29-39.
6. Vonderhaar BK. Prolactin: the forgotten hormone of human breast cancer. Pharmacol Ther 1998;79:169-78.
7. Bonneterre J, Peyrat JP, Beuscart R, Demaille A. Biological and clinical aspects of prolactin receptors in human breast cancer. J Steroid Biochem Mol Biol 1990;37:977-81.
8. Holtkamp W, Nagel GA, Wander HE, Rauschecker HF, von Heyden D. Hyperprolactinemia is a indicator of progressive disease and poor prognosis in advanced breast cancer. Int J Cancer 1984;34:323-8.
9. Patel DD, Bhatavdekar JM, Chikhlikar PR, Ghosh N, Suthar TP, Shah NG, et al. Node negative breast carcinoma: hyperprolactinemia and/or overexpression of p53 as an independent predictor of poor prognosis compared to newer and established prognosticators. J Surg Oncol 1996;62:83-92.
10. Bhatavdekar JM, Patel DD, Shah NG, Vora HH, Suthar TP, Ghosh N, et al. Prolactin as a local growth promoter in patients with breast cancer: GCRI experience. Eur J Surg Oncol 2000;26:540-7.
11. Love RR, Rose DR, Surawicz TS, Newcomb PA. Prolactin and growth hormone levels in premenopausal women with breast cancer and healthy women with a strong family history of breast cancer. Cancer 1991;68:1401-5.
12. Lachelin GCL, Yen SSC, Alksne JFN. Hormonal changes following hypophysectomy in humans. Obstet Gynecol 1977;50:333-9.
13. Manni A, Pearson OH, Brodkey J, Marshall JS. Transsphenoidal hypophysectomy in breast cancer: evidence for an individual role of pituitary and gonadal hormones in supporting tumor growth. Cancer 1979;44:2330-7.
14. Nolin JM, Witorsch RJ. Detection of endogenous immunoreactive prolactin in rat mammary epithelial cells during lactation. Endocrinology 1976;99:949-58.
15. Anderson E, Ferguson JE, Morten H, Shalet SM, Robinson EL, Howell A. Serum immunoreactive and bioactive lactogenic hormones in advanced breast cancer patients treated with bromocriptine and octreotide. Eur J Cancer 1993;29A:209-17.
16. Fields K, Kulig E, Lloyd RV. Detection of prolactin messenger RNA in mammary and other normal and neoplastic tissues by polymerase chain reaction. Lab Invest 1993;68:354-60.
17. Ginsburg E, Vonderhaar BK. Prolactin synthesis and secretion by human breast cancer cells. Cancer Res 1995;55:2591-5.
18. Clevenger CV, Chang W-P, Ngo W, Pasha TLM, Montone KT, Tomaszewski JE. Expression of prolactin and prolactin receptor in human breast carcinoma: evidence for an autocrine/paracrine loop. Am J Pathol 1995;146:695-705.
19. Kurtz A, Bristol LA, Toth BE, Lazar-Wesley E, Takacs L, Kacsoh B. Mammary epithelial cells of lactating rats express prolactin messenger ribonucleic acid. Biol Reprod 1993;48:1095-103.
20. Steinmetz RW, Grant AL, Malven PV. Transcription of prolactin gene in milk secretory cells of the rat mammary gland. J Endocrinol 1993;136:271-6.
21. Sinha YN. Structural variants of prolactin: occurrence and physiological significance. Endocr Rev 1995;16:354-69.
22. Clevenger CV, Russell DH, Appasamy PM, Prystowsky MB. Regulation of IL-2-driven T-lymphocyte proliferation by prolactin. Proc Natl Acad Sci USA 1990;87:6460-4.
23. Gellerson R, Kempf R, Teglmann R, DiMattia GE. Nonpituitary human prolactin gene transcription is indenpendent of pit-1 and differentially controlled in lymphocytes and in endometrial stroma. Mol Endocrinol 1994;8:356-73.
24. Richards RG, Hartman SM. Human dermal fibroblast cells express prolactin in vitro. J Invest Dermatol 1996;106:1250-5.
25. Ginsburg E, Das R, Vonderhaar BK. Prolactin: an autocrine growth factor in the mammary gland. In: Wilde CJ, Peaker M, Taylor E, eds. Biological Signalling in the Mammary Gland. Ayr, Scotland: Hannah Research Institute; 1997;47-58.
26. Reynolds C, Montone KT, Powell CM, Tomaszewski JE, Clevenger CV. Distribution of prolactin and its receptor in human breast carcinoma. Endocrinology 1997;138:5555-60.
27. Touraine P, Martini JF, Zafrani B, Durand JC, Labaille F, Malet C, et al. Increased expression of prolactin receptor gene assessed by quantitative polymerase chain reaction in human breast tumors versus normal breast tissue. J Clin Endocrinol Metab 1998;83:667-74.
28. Bazan JF. Structural design and molecular evolution of a cytokine receptor superfamily. Proc Natl Acad Sci USA 1990;87:6934-8.
29. Kelly PA, Djiane J, Postel-Vinay MC, Edery M. The prolactin/growth hormone receptor family. Endocr Rev 1991;12:235-51.
30. Kelly PA, Ali S, Rozakis M, Goujon L, Nagano M, Pellergini I, et al. The growth hormone/prolactin receptor family. Recent Prog Horm Res 1993;48:123-64.
31. Kline JB, Clevenger CV. Identification and characterization of the prolactin-binding protein (PRLBP) in human serum and milk. J Biol Chem 2001;276:24760-6.
32. Clevenger CV, Furth PA, Hankinson SE, Schuler LA. The role of prolactin in mammary carcinoma. Endocr Rev 2003;24:1-27.
33. Das R, Vonderhaar BK. Transduction of prolactin’s growth signal through both the long and short forms of the prolactin receptor. Mol Endocrinol 1995;9:1750-9.
34. Vanderhaar BK. Prolactin involvement in breast cancer. Endocrine-Related Cancer 1999;6:389-404.
35. Gill S, Peston D, Vonderhaar BK, Shousha S. Expression of prolactin receptors in normal, benign, and malignant breast tissue: an immunohistological study. J Clin Pathol 2001;54:956-60.
36. Ali S, Edery M, Pellegrini I, Lesueur L, Paly J, Djiane J, Kelly PA. The Nb2 form of prolactin receptor is able to activate a milk protein gene promoter. Mol Endocrinol 1992;6:1242-8.
37. Mertani HC, Garcia-Caballero T, Lambert A, Gerard F, Palayer C, Boutin JM, et al. Cellular expression of growth hormone and prolactin receptors in human breast disorders. Int J Cancer 1998;79:201-11.
38. Ormandy CJ, Hall RE, Manning DL, Robertson JFR, Blamey RW, Kelly PA, et al. Coexpression and cross-regulation of the prolactin receptor and sex steroid hormone receptors in breast cancer. J Clin Endocrinol Metab 1997;82:3692-9.
39. Plotnikov A, Li Y, Tran TH, Tang W, Palazzo JP, Rui H, Fuchs SY. Oncogene-mediated inhibition of glycogen synthase kinase 3 beta impairs degradation of prolactin receptor. Cancer Res 2008;68:1354-61.
40. Maus MV, Reilly SC, Clevenger CV. Prolactin as a chemoattractant for human breast carcinoma. Endocrinology 1999;140:5447-50.
41. Miller SL, Antico G, Raghunath PN, Tomaszewski JE, Clevenger CV. Nek3 kinase regulates prolactin-mediated cytoskeletal reorganization and motility of breast cancer cells. Oncogene 2007;26:4668-78.
42. Struman I, Bentzien F, Lee H, Mainfroid V, D’Angelo G, Goffin V, et al. Opposing actions of intact and N-terminal fragments of the human prolactin/growth hormone family members on angiogenesis: an efficient mechanism for the regulation of angiogenesis. Proc Natl Acad Sci USA 1999;96:1246-51.
43. Ferrara N, Clapp C, Weiner R. The 16K fragment of prolactin specifically inhibits basal or fibroblast growth factor stimulated growth of capillary endothelial cells. Endocrinology 1991;129:896-900.
44. Clapp C, Martial JA, Guzman RC, Rentier-Delrue F, Weiner RI. The 16-kilodalton N-terminal fragment of human prolactin is a potent inhibitor of angiogenesis. Endocrinology 1993;133:1292-9.
45. D’Angelo G, Martini JF, Iiri T, Fantl WJ, Martial J, Weiner RI. 16K human prolactin inhibits vascular endothelial growth factor-induced activation of Ras in capillary endothelial cells. Mol Endocrinol 1999;13: 692-704.
46. Lee H, Struman I, Clapp C, Martial J, Weiner RI. Inhibition of urokinase activity by the antiangiogenic factor 16K prolactin: activation of plasminogen activator inhibitor 1 expression. Endocrinology 1998;139:3696-703.
47. Clapp C, Weiner R. A specific, high affinity, saturable binding site for the 160 kilodalton fragment of prolactin on capillary endothelial cells. Endocrinology 1992;130:1380-6.
48. Goldhar AS, Vonderhaar BK, Trott JF, Hovey RC. Prolactin-induced expression of vascular endothelial growth factor via Erg-1. Mol Cell Endocrinol 2005;232:9-19.
49. Clapp C, Thebault S, de la Escalera GM. Role of prolactin and vasoinhibins in the regulation of vascular function in mammary gland. J Mammary Gland Biol Neoplasia 2008;13:55-67.
50. Rane SG, Reddy EP. Janus kinases: components of multiple signaling pathways. Oncogene 2000;19:5662-79.
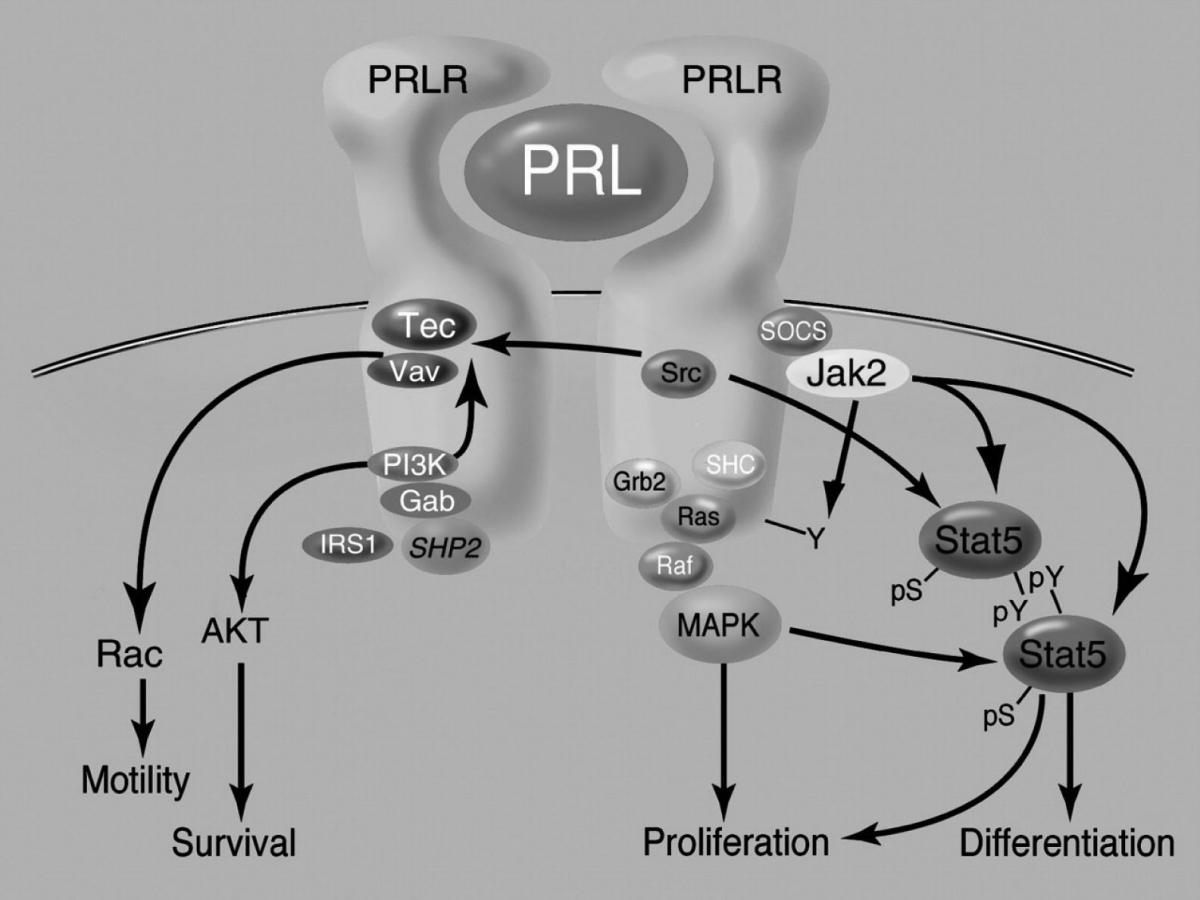
51. Clevenger CV, Kline JB. Prolactin receptor signal transduction. Lupus 2001;10:706-18.
52. Yu-Lee LY, Luo GY, Book ML, Morris SM. Lactogenic hormone signal transduction. Biol Reprod 1998;58:295-301.
53. Bole-Feysot C, Goffin V, Edery M, Binart N, Kelly PA. Prolactin (PRL) and its receptor: actions, signal transduction pathways and phenotypes observed in PRL receptor knockout mice. Endocr Rev 1998;19:225-68.
54. Fang F, Antico G, Zheng J, Clevenger CV. Quantification of PRL/STAT 5 signaling with a novel pGL4-CISH reporter. BMC Biotechnology 2008;8:11.
55. Darnell Jr JE, Kerr IM, Stark GR. Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 1994;264:1415-21.
56. DaSilva L, Rui H, Erwin RA, Howard OMZ, Kirken RA, Malabarba MG, et al. Prolactin recruits STAT1, STAT3, and STAT5 independent of conserved receptor tyrosine TYR402, TYR479, TYR515 and TYR 580. Mol Cell Endocrinol 1996;117:131-40.
57. Watson CJ, Miller WR. Elevated levels of members of the STAT family of transcription factors in breast carcinoma nuclear extracts. Br J Cancer 1995;71:840-4.
58. Bowman T, Garcia R, Turkson J, Jove R. STATs in oncogenesis. Oncogene 2000;19:2474-88.
59. Brockman JL, Schroeder MD, Schuler LA. Prolactin activates the cyclin D1 promoter via the JAK2-STAT pathway. Mol Endocrinol 2002;16:774-84.
60. Kazansky AV, Rosen JM. Signal transducers and activators of transcription 5B potentiate v-Src-mediated transformation of NIH-3T3 cells. Cell Growth Differ 2001;12:1-7.
61. Liu X, Robinson GW, Gouilleux F, Groner B, Henninghausen L. Cloning and expression of Stat5 and an additional homologue (Stat 5b) involved in prolactin signal transduction in mouse mammary tissue. Proc Natl Acad Sci USA 1995;92:8831-5.
62. Kazansky AV, Kabotyanski EB, Wsyzomierski SL, Mancini MA, Rosen JM Differential effects of prolactin and src/abl kinases on the nuclear translocation of STAT5B and STAT5A. J Biol Chem 1999;274:22484-92.
63. Berlanga JJ, Vara JAF, Martín-Pérez J, García-Ruiz JP. Prolactin receptor is associated with c-src kinase in rat liver. Mol Endocrinol 1995;9:1461-7.
64. Fang F, Rycyzyn MA, Clevenger CV. Role of c-Myb during prolactin-induced signal transducer and activator of transcription 5a signaling in breast cancer cells. Endocrinology 2009;150:1597-606.
65. Swaminathan G, Varghese B, Thangavel C, Carbone CJ, Plotnikov A, Kumar KG, et al. Prolactin stimulates ubiquitination, initial internalization, and degradation of its receptor via catalytic activation of Janus kinase 2. Endocrinology 2008;196:R1-7.
66. Clevenger CV, Torigoe T, Reed JC. Prolactin induces rapid phosphorylation and activation of prolactin receptor associated Raf-1 kinase in a T-cell line. J Biol Chem 1994;269:5559-65.
67. Llovera M, Pichard C, Bernichtein S, Jeay S, Touraine P, Kelly PA, Goffin V. Human prolactin (hPRL) antagonists inhibit hPRL-activated signaling pathways involved in breast cancer cell proliferation. Oncogene 2000;19:4695-705.
68. Das R, Vonderhaar BK. Activation of raf-1 MEK, and MAP kinase in prolactin responsive mammary cells. Breast Cancer Res Treat 1996;40:141-9.
69. Das R, Vonderhaar BK. Involvement of SHC, GRB2, SOS and RAS in prolactin signal transduction in mammary epithelial cells. Oncogene 1996;13:1139-45.
70. Yamauchi T, Yamauchi N, Ueki K, Sugiyama T, Waki H, Miki H, et al. Constitutive tyrosine phosphorylation of ErbB-2 via Jak2 by autocrine secretion of prolactin in human breast cancer. J Biol Chem 2000;275:33937-44.
71. Gao J, Horseman ND. Prolactin-independent modulation of the ß-casein response element by Erk2 MAP kinase. Cell Signal 1999;11:205-10.
72. Decker T, Kovarik P. Serine phosphorylation of STATs. Oncogene 2000;19:2628-37.
73. Roymans D, Slegers H. Phosphatidylinositol 3-kinases in tumor progression. Eur J Biochem 2001;268:487-98.
74. Blume-Jensen P, Hunter T. Oncogenic kinase signalling. Nature 2001; 11:355-65.
75. Cantrell DA. Phosphoinositide 3-kinase signalling pathways. J Cell Sci 2001;114:1439-45.
76. Rodriguez-Viciana P, Marte BM, Warne PH, Downward J. Phosphatidylinositol 3’ kinase: one of the effectors of Ras. Philos Trans R Soc Lond B Biol Sci 1996;351:225-31.
77. Constantino S, Santos R, Lacronique V, Bouchaert I, Monni R, Bernard O, et al. Constitutively active STAT5 variants induce growth and survival of hematopoietic cells through a PI 3-kinase/Akt dependent pathway. Oncogene 2001;20:2080-90.
78. Craddock BL, Hobbs J, Edmead CE, Welham MJ. Phosphoinositide 3-kinase-dependent regulation of interleukin-3-induced proliferation: involvement of mitogen-activated protein kinases, SHP2 and Gab2. J Biol Chem 2001;276:24274-83.
79. Kline JB, Moore DJ, Clevenger CV. Activation and association of the Tec tyrosine kinase with the human prolactin receptor: mapping of a Tec/Vav1-receptor binding site. Mol Endocrinol 2001;15:832-41.
80. Clevenger CV, Ngo W, Luger SM, Gewirtz AM. Vav is necessary for prolactin-stimulated proliferation and is translocated into the nucleus of a T-cell line. J Biol Chem 1995;270:13246-53.
81. Clevenger CV, Altmann SW, Prystowsky MB. Requirement of nuclear prolactin for interleukin-2-stimulated proliferation of T lymphocytes. Science 1991;253:77-9.
82. Rycyzyn MA, Reilly SC, O’Malley K, Clevenger CV. Role of cyclophilin B in PRL signal transduction and nuclear retrotranslocation. Mol Endocrinol 2000;14:1175-86.
83. Rycyzyn MA, Clevenger CV. The intranuclear prolactin/cyclophilin B complex as a transcriptional inducer. Proc Natl Acad Sci USA 2002;99:6790-5.
84. Vonderhaar BK, Biswas R. Prolactin effects and regulation of its receptors in human mammary tumor cells. In: Medina D, Kidwell W, Hepner G, Anderson E, eds. Cellular and Molecular Biology of Mammary Cancer. New York: Plenum Publishing Corp; 1987;205-19.
85. Welsch CW, Gribler C. Prophylaxis of spontaneously developing mammary carcinoma in C3H/HeJ female mice by suppression of prolactin. Cancer Res 1973;33:2939-46.
86. Welsch CW. Prolactin and the development and progression of early neoplastic mammary gland lesions. Cancer Res 1978;38:4054-8.
87. Welsch CW, Goodrich-Smith M, Brown CK, Roth L. The prophylaxis of rat and mouse mammary gland tumorigenesis by suppression of prolactin secretion: a reappraisal. Breast Cancer Res Treat 1981;1:225-32.
88. Wennbo H, Gebre-Medhin M, Griti-Linde A, Ohlsson C, Isaksson OGP, Tornell J. Activation of the prolactin receptor but not the growth hormone receptor is important for induction of mammary tumors in transgenic mice. J Clin Invest 1997;100:2744-51.
89. Wennbo H, Tornell J. The role of prolactin and growth hormone in breast cancer. Oncogene 2000;19:1072-6.
90. Thordarson G, Van Horn K, Guzman RC, Nandi S, Talamantes F. Parous rats regain high susceptibility to chemically induced mammary cancer after treatment with various mammotropic hormones. Carcinogenesis 2001;22:1027-33.
91. Wang DY, Stepniewska KA, Allen DS, Fentiman IS, Bulbrook RD, Kwa HG, et al. Serum prolactin levels and their relationship to survival in women with operable breast cancer. J Clin Epidemiol 1995;48:959-68.
92. Lissoni P, Barni S, Cazzaniga M, Ardizzoia A, Rovelli F, Tancini G, et al. Prediction of recurrence in operable breast cancer by postoperative changes in prolactin secretion. Oncology 1995;52:439-42.
93. Bhatavdekar JM, Shah NG, Balar DB, Patel DD, Bhaduri A, Trivedi SN, et al. Plasma prolactin as an indicator of disease progression in advanced breast cancer. Cancer 1990;65:2028-32.
94. Wang DY, De Stavola BL, Bulbrook RD, Allen DS, Kwa HG, Verstraeten AA, et al. The permanent effect of reproductive events on blood prolactin levels and its relation to breast cancer risk: a population study of postmenopausal women. Eur J Cancer Clin Oncol 1988;24:1225-31.
95. Ingram DM, Nottage EM, Roberts AN. Prolactin and breast cancer risk. Med J Aust 1990;153:469-73.
96. Hankinson SE, Colditz GA, Hunter DJ, Manson JE, Willett WC, Stampfer MJ, et al. Reproductive factors and family history of breast cancer in relation to plasma estrogen and prolactin levels in postmenopausal women in the Nurses’ Health Study (United States). Cancer Causes Control 1995;6:217-24.
97. Boyd NF, Stone J, Martin L, Minkin S, Yaffe M. Mammographic densities and the growth hormone-IGF-1 prolactin axis. Sunnybrook and Women’s College Hospital, Proc 92nd Meeting of the American Association for Cancer Research, New Orleans, LA, 2001;558.
98. Martini MC, Dancisak BB, Haggans CJ, Thomas W, Slavin JL. Effects of soy intake on sex hormone metabolism in premenopausal women. Nutr Cancer 1999;34:133-9.
99. Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53,297 women with breast cancer and 100,239 women without breast cancer from 54 epidemiological studies. Lancet 1996;347:3106-8.
100. Rossing MA, Scholes D, Cushing-Haugen KL, Voigt LF. Cimetidine use and risk of prostate and breast cancer. Cancer Epidemiol Biomarkers Prev 2000;9:319-23.
101. Strungs I, Gray RA, Rigby HB, Strutton G. Two case reports of breast carcinoma associated with prolactinoma. Pathology 1997;29:320-3.
102. Volm MD, Talamonti MS, Thangavelu M, Gradishar WK. Pituitary adenoma and bilateral male breast cancer: an unusual association. J Surg Oncol 1997;64:74-8.
103. Popovic V, Damjanovic S, Micic D, Nesovic M, Djurovic M, Petakov M, et al. Increased incidence of neoplasia in patients with pituitary adenomas. The Pituitary Study Group. Clin Endocrinol (Oxf) 1998;49:441-5.
104. Tworoger SS, Sluss P, Hankinson SE. Association between plasma prolactin concentrations and risk of breast cancer among predominately premenopausal women. Cancer Res 2006;66:2476-82.
105. Lee SA, Haiman CA, Burtt NP, Pooler LC, Cheng I, Kolonel LN, Pike MC, Altshuler D, Hirschhorn JN, Henderson BE, Stram DO. A comprehensive analysis of common genetic variations in prolactin (PRL) and PRL receptor (PRLR) genes in relation to plasma prolactin levels and breast cancer risk: the multiethnic cohort. BMC Med Genet 2007;8:72.
106. Holtkamp W, von Heyden D, Rauschecker H, Nagel GA. Plasma-prolactin concentrations in breast cancer at various stages, in mastopathy and other malignant tumors. Schweiz Med Wochenschr 1983;113:1513-20.
107. Holtkamp W, Wuttke W, Nagel GA, Michel U, Rauschecker H. Pathophysiology of hyperprolactinemia in breast cancer. Onkologie 1988;11:86-103.
108. Bhatavdekar JM, Patel DD, Vora HH, Ghosh N, Shah NG, Karelia NH, et al. Node-positive breast cancer: prognostic significance of the plasma prolactin compared with steroid receptors and clinicopathological features. Oncology Reports 1994;1:841-5.
109. Bhatavdekar JM, Patel DD, Shah NG, Vora HH, Suthar TP, Chikhlikar PR, et al. Prognostic significance of immunohistochemically localized biomarkers in stage II and stage III breast cancer: a multivariate analysis. Ann Surg Oncol 2000;7:305-11.
110. Bhatavdekar JM, Patel DD, Shah NG, Vora HH, Suthar TP, Ghosh N, et al. Prolactin as a local growth promoter in patients with breast cancer: GCRI experience. Eur J Surg Oncol 2000;26:540-7.
111. Mujagić Z, Mujagić H. Diagnostic and prognostic usefulness of prolactin (PRL) in breast cancer (BC). Proceedings of ASCO, San Francisco 2001;20:43b.
112. Mujagić Z, Mujagić H. Circulating levels of prolactin as an indicator of effectiveness of therapy in breast cancer patients. Int J Cancer 2002; Suppl 13:216.
113. Mujagić Z, Mujagić H. Prognostic significance of circulating levels of prolactin (PRL) in metastatic breast cancer (MBC). Proceedings of ASCO, Chicago 2003;22:49.
114. Mujagić Z, Mujagić H. Importance of serum prolactin determination in metastatic breast cancer patients. Croat Med J 2004;45:176-80.
115. Mujagić Z, Mujagić H. The relationship of circulating prolactin levels to the size of primary tumor in breast cancer patients. Turk J Bioch 2004;29:277-81.
116. Mandala M, Lissoni P, Ferretti G, Rocca A, Torri V, Moro C, et al. Postoperative hyperprolactinemia could predict longer disease-free and overall survival in node-negative breast cancer patients. Oncology 2002;63:370.
117. Gomez F, Reyes FI, Faiman C. Nonpuerperal galactorrhea and hyperprolactinemia. Clinical findings, endocrine features and therapeutic responses in 56 cases. Am J Med 1977;62:648-60.
118. Berinder K, Stackenäs I, Akre O, Hirschberg AL, Hulting AL. Hyperprolactinemia in 271 women: up to three decade of clinical follow-up. Clin Endocrinol (Oxf) 2005;63:450-5.
119. Kadioglu P, Yalin AS, Tiryakioglu O, Gazioglu N, Oral G, Sanli O, Onem K, Kadioglu A. Sexual dysfunction in women with hyperprolactinemia: a pilot study report. J Urol. 2006;176:841-2.
120. Suba Z. Carcinogenesis theory based on estrogen deficiency. Orv Hetil 2009;150:1155-6.
121. Plotnikov A, Varghese B, Tran TH, Liu C, Rui H, Fuchs SY. Impaired turnover of prolactin receptor contributes to transformation of human breast cells. Cancer Res 2009;69:OF1-8.
122. Vonderhaar BK, Banerjee R. Is tamoxifene also an antilactogen? Mol Cell Endocrinol 1991;79:C159-C163.
123. Das R, Biswas R, Vonderhaar BK. Characteristics of the antilactogen binding site in mammary gland membranes. Mol Cell Endocrinol 1993;98:1-8.
124. Sutherland RL, Murphy LC, Foo MS, Green MD, Whybourne AM, Krozowski ZS. High-affinity anti-oestrogen binding sites distinct from the oestrogen receptor. Nature 1980;288:273-5.
125. Das R, Vonderhaar BK. Prolactin as a mitogen in mammary cells. J Mammary Gland Biol Neoplasia 1997;2:29-39.
126. Das R, Vonderhaar BK. Tamoxifen inhibits prolactin signal transduction in estrogen receptor negative NOG-8 mammary epihelial cells. Cancer Letters 1997;116:41-6.
127. Faupel-Badger JM, Prindiville SA, Venzon D, Vonderhaar BK, Zujewski JA, Eng-Wong J. Effects of raloxifene on circulating prolactin and estradiol levels in premenopausal women at high risk for developing breast cancer. Cancer Epidemiol Biomarkers Prev 2006;15:1153-8.