References
1. Olsson I, Venge P. Cationic proteins of human granulocytes. II Separation of the cationic proteins of the granules of leukemic myeloid cells. Blood 1974;44:235-46.
2. Venge P, Byström J, Carlson M, Håkansson L, Karawacjzyk M, Peterson C, et al. Eosinophil cationic protein (ECP): molecular and biological properties and the use of ECP as a marker of eosinophil activation in disease. Clin Exp Allergy 1999;29:1172-86.
3. Zhang J, Rosenberg HF. Sequence variation at two eosinophil-associated ribonuclease loci in humans. Genetics 2000;156:1949-58.
4. Trulson A, Byström J, Engström A, Larsson R, Venge P. The functional heterogeneity of eosinophil cationic protein is determined by a gene polymorphism and post-translational modifications. Clin Exp Allergy 2007;37:208-18.
5. Eriksson J, Woschnagg C, Fernvik E, Venge P. A SELDI-TOF MS study of the genetic and post-translational molecular heterogeneity of eosinophil cationic protein. J Leukocyte Biol 2007;82:1491-1500.
6. Byström J, Garcia RC, Håkansson L, Karawajczyk M, Moberg L, Soukka J, Venge P. Eosinophil cationic protein is stored in, but not produced by peripheral blood neutrophils. Clin Exp Allergy 2002;32:1082-91.
7. Byström J, Tenno T, Håkansson L, Amin K, Trulson A, Högbom E, Venge P. Monocytes but not macrophages produced the eosinophil cationic protein. APMIS 2001;109:507-16.
8. Peters MS, Rodriquez M, Gleich GJ. Localisation of human eosinophil granule major basic protein, eosinophil cationic protein and eosinophil derived neurotoxin by immunoelectron microscopy. Lab Invest 1986;54:656-62.
9. Egesten A, Calafat J, Weller PF, Knol EF, Janssen H, Walz TM, Olsson I. Localization of granule proteins in human eosinophil bone marrow progenitors. Int Arch Allergy Immunol 1997;114:130-8.
10. Monteseirín J, Vega A, Chacón P, Camacho MJ, El Bekay R, Asturias JA, et al. Neutrophils as a novel source of eosinophil cationic protein in IgE-mediated processes. J Immunol 2007;179:2634-41.
11. Tomassini M, Tsicopoulos A, Tai PC, Gruart V, Tonnel A-B, Prin L, et al. Release of granule protein by eosinophils from allergic and nonallergic patients with eosinophilia on immunoglobulin-dependent activation. J Allergy Clin Immunol 1991;88:365-75.
12. Carlson M, Peterson C, Venge P. The influence of IL-3, IL-5 and GM-CSF on normal human eosinophil and neutrophil C3b-induced degranulation. Allergy 1993;48:437-42.
13. Erjefält JS, Greiff L, Andersson M, Matsson E, Petersen H, Linden M, et al. Allergen-induced eosinophil cytolysis is a primary mechanism for granule protein release in human upper airways. Am J Respir Crit Care Med 1999;160:304-12.
14. Neves JS, Weller PF. Functional extracellular eosinophil granules: novel implications in eosinophil immunobiology. Curr Opin Immunol 2009;21:694-9.
15. Woschnagg C, Rubin J, Venge P. Eosinophil cationic protein (ECP) is processed during secretion. J Immuno 2009;183:3949-54.
16. Meng Q, Ying S, Corrigan CJ, Wakelin M, Assoufi B, Moqbel R, Kay AB. Effect of rapamycin, cyclosporin A and dexamethasone on interleukin 5-induced eosinophil degranulation and prolonged survival. Allergy 1997;52:1095-101.
17. Eda R, Sugiyama H, Hopp RJ, Okada C, Bewtra AK, Townley RG. Inhibitory effects of formoterol on platelet-activating factor induced eosinophil chemotaxis and degranulation. Int Arch Allergy Immunol 1993;102:391-8.
18. Kawashima T, Iwamoto I, Nakagawa N, Tomioka H, Yoshida S. Inhibitory effect of pemirolast, a novel antiallergic drug, on leukotriene C4 and granule protein release from human eosinophils. Int Arch Allergy Immunol 1994;103:405-9.
19. Eriksson J, Reimert CM, Kabatereine NB, Kazibwe F, Ireri E, Kadzo H, et al. The 434(G>C) polymorphism within the coding sequence of eosinophil cationic protein (ECP) correlates with the natural course of Schistosoma mansoni infection. Int J Parasitol 2007;37:1359-66.
20. Rubin J, Zagai U, Blom K, Trulson A, Engström A, Venge P. The coding ECP 434(G>C) gene polymorphism determines the cytotoxicity of ECP but has minor effects on fibroblast-mediated gel contraction and no effect on RNase activity J Immunol 2009;183:445-51.
21. Jönsson U-B, Håkansson L, Jögi R, Janson C, Venge P. Associations of ECP (eosinophil cationic protein) - gene polymorphisms to allergy, asthma, smoke habits and lung function in two Estonian and Swedish sub cohorts of the ECRHS II study. BMC Pulm Med 2010:10:36.
22. Jönsson U-B, Byström J, Stålenheim G, Venge P. Polymorphism of the eosinophil cationic protein-gene is related to the expression of allergic symptoms. Clin Exp Allergy 2002;32:1092-5.
23. Legrand F, Driss V, Delbeke M, Loiseau S, Hermann E, Dombrowicz D, Capron M. Human eosinophils exert TNF-alpha and granzyme A-mediated tumoricidal activity toward colon carcinoma cells. J Immunol 2010;185:7443-51.
24. Ishibashi S, Ohashi Y, Suzuki T, Miyazaki S, Moriya T, Satomi S, Sasano H. Tumor-associated tissue eosinophilia in human esophageal squamous cell carcinoma. Anticancer Res 2006;26:1419-24.
25. Oliveira DT, Tjioe KC, Assao A, Faustino SES, Carvalho AL, Landman G, Kowalski LP. Tissue eosinophilia and its association with tumoral invasion of oral cancer. Int J Surg Pathol 2009;17:244-9.
26. Molin D, Glimelius B, Sundström C, Venge P, Enblad G. The serum levels of eosinophil cationic protein (ECP) are related to the infiltration of eosinophils in the tumours of patients with Hodgkin’s disease. Leuk Lymphoma 2001;42:457-65.
27. Martinelli-Kläy CP, Mendis BRRN, Lombardi T. Eosinophils and oral squamous cell carcinoma: A short review. J Oncology 2009;2009:310132.
28. Pereira MC, Oliveira DT, Olivieri EHR, Rogatto SR, Carvalho AL, Landman G, Kowalski LP. The 434(G>C) polymorphism in the eosinophil cationic protein gene and its association with tissue eosinophilia in oral squamous cell carcinomas. J Oral Pathol Med 2010;39:56-62.
29. Jönssson U-B, Byström J, Stålenheim G, Venge P. A (G>C) transversion in the 3’ UTR of the human ECP (eosinophil cationic protein) gene correlates to the cellular content of ECP. J Leukocyte Biol 2006;79:846-51.
30. Kozak M. How strong is the case for regulation of the initiation step of translation by elements at the 3’ end of eukaryotic mRNAs? Gene 2004;343:41-54.
31. Munthe-Kaas MC, Gerritsen J, Carlsen KH, Undlien D, Egeland T, Skinningsrud B, et al. Eosinophilic cationic protein (ECP) polymorphisms and association with asthma, s-ECP levels and related phenotypes. Allergy 2007;62:429-36.
32. McLaren DJ, Peterson CG, Venge P. Schistosoma mansoni: further studies of the interaction between schistosomula and granulocyte-derived cationic proteins in vitro. Parasitol 1984;88:491-503.
33. Lehrer RI, Szklarez D, Barton A, Ganz T, Hamann KJ, Hleich GJ. Anti-bacterial properties of eosinophil major basic protein (MBP) and eosinophil cationic protein (ECP). J Immunol 1989;142:4428-34.
34. Domachowske JB, Dyer KD, Adams AG, Leto TL, Rosenberg HF. Eosinophil cationic protein/RNase 3 is another RNase A-family ribonuclease with direct antiviral activity. Nucl Acids Res 1998;26:3358-63.
35. Boix E, Torrent M, Sánchez D, Nogués MV. The antipathogen activities of eosinophil cationic protein. Curr Pharm Biotechnol 2008;9:141-52.
36. Young JD, Peterson CG, Venge P, Cohn ZA. Mechanism of membrane damage mediated by human eosinophil cationic protein. Nature 1986;321:613-6.
37. Miner JH. Laminins and their roles in mammals. Microsc Res Tech 2008;71:349-56.
38. Torrent M, Odorizzi F, Nogués MV, Boix E. Eosinophil cationic protein aggregation: identification of an N-terminus amyloid prone region. Biomacromolecules 2010;11:1983-90.
39. Torrent M, Badia M, Moussaoui M, Sanchez D, Nogués MV, Boix E. Comparison of human RNase 3 and RNase 7 bactericidal action at the Gram-negative and Gram-positive bacterial cell wall. FEBS Journal 2010;277:1713-25.
40. Klion AD, Nutman TB. The role of eosinophils in host defense against helminth parasites. J Allergy Clin Immunol 2004;113:30-7.
41. Zagai U, Lundahl J, Klominek J, Venge P, Sköld CM. Eosinophil cationic protein stimulates migration of human lung fibroblasts in vitro. Scand J Immunol 2010;69:381-6.
42. Rosenberg HF, Domachowske JB. Eosinophils, eosinophil ribonucleases and their role in host defense against respiratory virus pathogens. J Leukoc Biol 2001;70:691-8.
43. Garofalo R, Kimpen JI, Welliver RC, Ogra PI. Eosinophil degranulation in the respiratory tract during naturally acquired respiratory syncytial virus infection. J Pediatr 1992;120:28-32.
44. Domachowske JB, Rosenberg HF. Respiratory syncytial virus infection: immune response, immunopathogenesis and treatment. Clin Microbiol Rev 1999;12:298-309.
45. Torrent M, De La Torre BG, Nogués VM, Andreu D, Boix E. Bactericidal and membrane disruption activities of the eosinophil cationic protein are largely retained in an N-terminal fragment. Biochem J 2009;421:425-34.
46. Torrent M, Navarro S, Moussaoui M, Nogués MV, Boix E. Eosinophil cationic protein high-affinity binding to bacteria-wall lipopolysaccharides and peptidoglycans. Biochemistry 2008;47:3544-55.
47. Trautmann A, Schmid-Grendelmeier P, Kruger K, Crameri R, Akdis M, Akkaya A, et al. T cells and eosinophils cooperate in the induction of bronchial epithelial cell apoptosis in asthma. J Allergy Clin Immunol 2002;109:329-37.
48. Navarro S, Boix E, Cuchillo CM, Nogués MV. Eosinophil-induced neurotoxicity: The role of eosinophil cationic protein/RNase 3. J Neuroimmun 2010;227:60-70.
49. Chang KC, Lo CW, Fan TC, Chang MDT, Shu CW, Chang CH, et al. TNF-α mediates eosinophilic cationic protein-induced apoptosis in BEAS-2B cells. BMC Cell Biol 2010;11:6.
50. Plager DA, Davis MDP, Andrews AG, Coenen MJ, George TJ, Gleich GJ, Leiferman KM. Eosinophil ribonucleases and their cutaneous lesion-forming activity. J Immunol 2009;183:4013-20.
51. Gomes I, Mathur SK, Espenshade BM, Mori Y, Varga J, Ackerman SJ. Eosinophil-fibroblast interactions induce fibroblast IL-6 secretion and extracellular matrix gene expression: Implications in fibrogenesis. J Allergy Clin Immunol 2005;116:796-804.
52. Niccoli G, Ferrante G, Cosentino N, Conte M, Belloni F, Marino M, et al. Eosinophil cationic protein: A new biomarker of coronary atherosclerosis. Atherosclerosis 2010;211:606-11.
53. Peona V, De Amici M, Quaglini S, Bellaviti G, Castellazzi AM, Marseglia G, Ciprandi G. Serum eosinophilic cationic protein: Is there a role in respiratory disorders? J Asthma 2010;47:131-4.
54. Koh GC-H, Shek LP-C, Goh DY-T, Van Bever H, Koh DS-Q. Eosinophil cationic protein: Is it useful in asthma? A systematic review. Respir Med 2007;101:696-705.
55. Kato M, Yamada Y, Maruyama K, Hayashi Y. Serum eosinophil cationic protein and 27 cytokines/chemokines in acute exacerbation of childhood asthma. Int Arch Allergy Immunol 2010;152(Suppl 1):62-6.
56. Kämpe M, Stolt I, Lampinen M, Janson C, Stålenheim G, Carlson M. Patients with allergic rhinitis and allergic asthma share the same pattern of eosinophil and neutrophil degranulation after allergen challenge. Clin Mol Allergy 2011;9:3.
57. Hogan SP, Rothenberg ME. Eosinophil function in eosinophil-associated gastrointestinal disorders. Curr Allergy Rep 2006;6:65-71.
58. Byström J, Amin K, Bishop-Bailey D. Analysing the eosinophil cationic protein - a clue to the function of the eosinophil granulocyte. Respiratory Research 2011;12:10.
59. Park YJ, Oh EJ, Park JW, Kim M, Han K. Plasma eosinophilic cationic protein, interleukin-5 and ECP/Eo count ratio in patients with various eosinophilic diseases. Ann Clin Lab Sci 2006;36:262-6.
60. Koller DY, Nilsson M, Enander I, Venge P, Eichler I. Serum eosinophil cationic protein, eosinophil protein X and eosinophil peroxidase in relation to pulmonary function in cystic fibrosis. Clin Exp Allergy 1998;28:241-8.
61. Peterson GB, Eklund E, Yesuf T, Yngve R, Carlson M. A new method for quantification of neutrophil and eosinophil cationic protein in feces: Establishment of normal levels and clinical application in patients with inflammatory bowel diseas. Am J Gastroenterol 2002;97:1755-61.
62. Venge P. Monitoring the allergic inflammation. Allergy 2004;59:26-32.
63. Joseph-Bowen J, de Klerk N, Holt PG, Sly PD. Relationship of asthma, atopy and bronchial responsiveness to serum eosinophil cationic proteins in early childhood. J Allergy Clin Immunol 2004;114:1040-5.
64. Matsumoto H, Niimi A, Minakuchi T, Izumi T. Serum eosinophil cationic protein levels measured during exacerbation of asthma: characteristics of patients with low titers. Clin Exp Allergy 2001;31:637-43.
65. Hyvärinen MK, Kotaniemi-Syrjänen A, Reijonen TM, Piippo-Savolainen E, Korppi M. Eosinophil activity in infants hospitalized for wheezing and risk of persistent childhood asthma. Pediatr Allergy Immunol 2010;21:96-103.
66. Reimert CM, Poulsen LK, Bindslev-Jensen C, Kharazmi A, Bendtzen K. Measurement of eosinophil cationic protein (ECP) and eosinophil protein X/eosinophil derived neurotoxin (EPX/EDN). J Immunol Meth 1993;166:183-90.
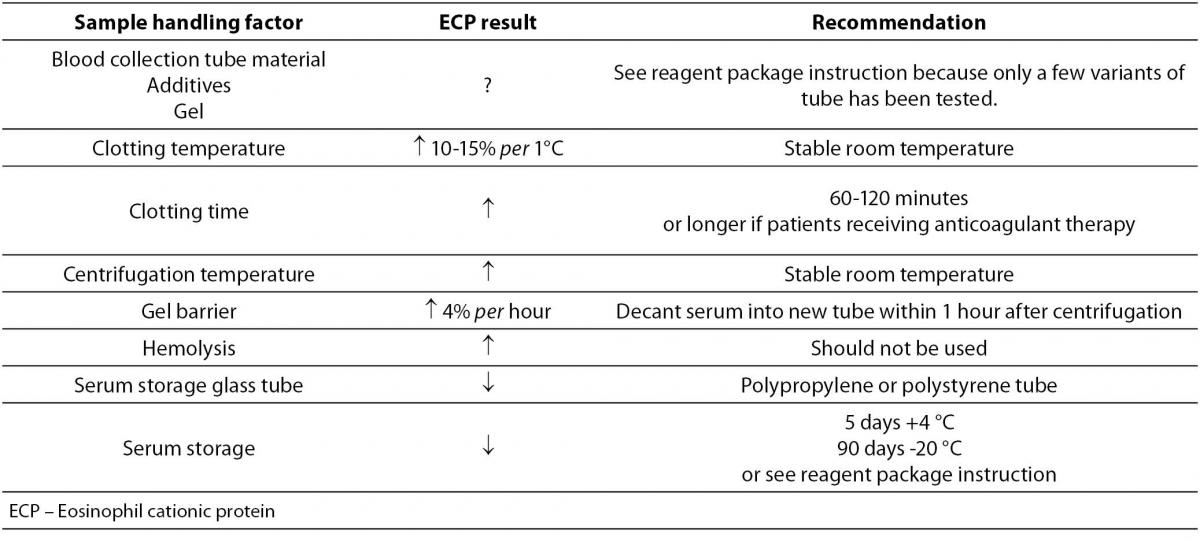
67. Venge AB, Peterson CGB. Measurements of ECP in serum and the impact of plasma coagulation. Allergy 2000;55:442-8.
68. Peterson CG, Venge P. Interaction and complex-formation between the eosinophil cationic protein and α2-macroglobulin. Biochem J 1987;245:781-7.
69. Pronk-Admiraal CJ, Bartles PC. Effect of clotting temperature and eosinophil concentration on the eosinophil cationic protein concentration in serum. Scand J Clin Lab Invest 1994;54:185-8.
70. Hoekstra MO, Grol MH, Hovenga H, Bouman K, Stijnen T, Koëter GH. Eosinophil and mast cell parameters in children with stable moderate asthma. Pediatr Allergy Immunol 1998;9:143-9.
71. Sjöswärd KN, Uppugunduri S, Schmekel B. Decreased serum levels of P-selectin and eosinophil cationic protein in patients with mild asthma after inhalated salbutamol. Respiration 2004;71:241-5.
72. Wolthers OD, Heuck C. Circadian variations in serum eosinophil cationic protein and serum and urine eosinophil protein X. Pediatr Allergy Immunol 2003;14:130-3.
73. Ferdousi HA, Munir AKM, Zetterström O, Dreborg SKG. Seasonal differences of peak expiratory flow rate variability and mediators of allergic inflammation in non-atopic adolescents. Pediatr Allergy Immunol 2001;12:238-46.
74. Haahtela T. Assessing airway inflamation: from guessing to quantitative measurements. Ann Med 2002;34:74-6.
75. Bartoli ML, Di Franco A, Vagaggini B, Bacci E, Cianchetti S, Dente FL, et al. Biological markers in induced sputum of patients with different phenotypes of chronic airway obstruction. Respiration 2009;77:265-72.
76. Metsto T, Venge P, Haahtela T, Peterson CG, Seveus L. Cell specific markers for eosinophils and neutrophils in sputum and bronchoalveolar lavage fluid of patients with respiratory conditions and healthy subjects. Thorax 2002;57:449-51.
77. Keatings VM, Jatakanon A, Worsdell YM, Barnes PJ. Effects of inhalated and oral glucocorticoids on inflammatory indices in asthma and COPD. Am J Respir Crit Care Med 1997;155:542-8.
78. Simpson JL, Timmins NL, Fakes K, Talbot PI, Gibson PG. Effect of saliva contamination on induced sputum cell count, IL-8 and eosinophil cationic protein levels. Eur Respir J 2004;23:759-62.
79. Kelly MM, Keatings V, Leigh R, Peterson C, Shute J, Venge P, et al. Analysis of fluid-phase mediators. Eur Respir J 2002;37:24-39.
80. Koh GC, Shek LP, Kee J, Wee A, Ng V, Koh D. Saliva and serum eosinophil cationic protein in asthmatic children and adolescents with and without allergic sensitization. J Asthma 2010;47:61-5.
81. Boot JD, Chandoesing P, de Kam ML, Mascelli MA, Das AM, van Wijk RG, et al. Applicability and reproducibility of biomarkers for the evaluation of anti-inflammatory therapy in allergic rhinitis. J Investig Allergol Clin Immunol 2008;18:433-42.
82. Lü FX, Esch RE. Novel nasal secretion collection method for the analysis of allergen specific antibodies and inflamatory markers. J Immunol Methods 2010;356:6-17.
83. Rondón C, Romero JJ, López S, Antúnez C, Martín-Casañez E, Torres MJ, et al. Local IgE production and positive nasal provocation test in patients with persistent nonallergic rhinitis. J Allergy Clin Immunol 2007;119:899-905.
84. Rondón C, Fernández J, López S, Campo P, Doña I, Torres MJ, et al. Nasal inflammatory mediators and specific IgE production after nasal challenge with grass pollen in local allergic rhinitis. J Alergy Clin Immunol 2009;124:1005-11.
85. Silva AC, Levy L, Trindade JC, Mendonça P, Silva C, Lopes AI. Faecal and serum levels of eosinophil cationic protein in a healthy paediatric population. Scand J Clin Lab Invest 2007;67:757-66.