References
1. Wallin O, Soderberg J, Van Guelpen B, Stenlund H, Grankvist K, Brulin C. Preanalytical venous blood sampling practices demand improvement--a survey of test-request management, test-tube labelling and information search procedures. Clin Chim Acta 2008;391:91-7.
2. Lippi G, Bassi A, Brocco G, Montagnana M, Salvagno GL, Guidi GC. Preanalytic error tracking in a laboratory medicine department: results of a 1-year experience. Clin Chem 2006;52:1442-3.
3. Carraro P, Plebani M. Errors in a stat laboratory: types and frequencies 10 years later. Clin Chem 2007;53:1338-42.
4. Plebani M, Carraro P. Mistakes in a stat laboratory: types and frequency. Clin Chem 1997;43:1348-51.
5. Lippi G, Fostini R, Guidi GC. Quality improvement in laboratory medicine: extra-analytical issues. Clin Lab Med 2008;28:285-94.
6. Lippi G, Guidi GC. Risk management in the preanalytical phase of laboratory testing. Clin Chem Lab Med 2007;45:720-7.
7. Lima-Oliveira GS, Picheth G, Sumita NM, Scartezini M. Quality control in the collection of diagnostic blood specimens: illuminating a dark phase of preanalytical errors. J Bras Patol Med Lab 2009;45:441-7.
8. Lippi G, Guidi GC. Preanalytic indicators of laboratory performances and quality improvement of laboratory testing. Clin Lab 2006;52:457-62.
9. Bilić-Zulle L, Šimundić A-M, Smolčić VŠ, Nikolac N, Honović L. Self reported routines and procedures for the extra-analytical phase of laboratory practice in Croatia -cross-sectional survey study. Biochem Med 2010;20:64-74.
10. Lippi G, Salvagno GL, Montagnana M, Franchini M, Guidi GC. Phlebotomy issues and quality improvement in results of laboratory testing. Clin Lab 2006;52:217-30.
11. CLSI. Procedures for the handling and processing of blood specimens for common laboratory tests. NCCLS H18-A4. 4 ed2010.
12. CLSI. Procedures for the collection of diagnostic blood specimens by venipuncture. NCCLS H3-A6. 6 ed2007.
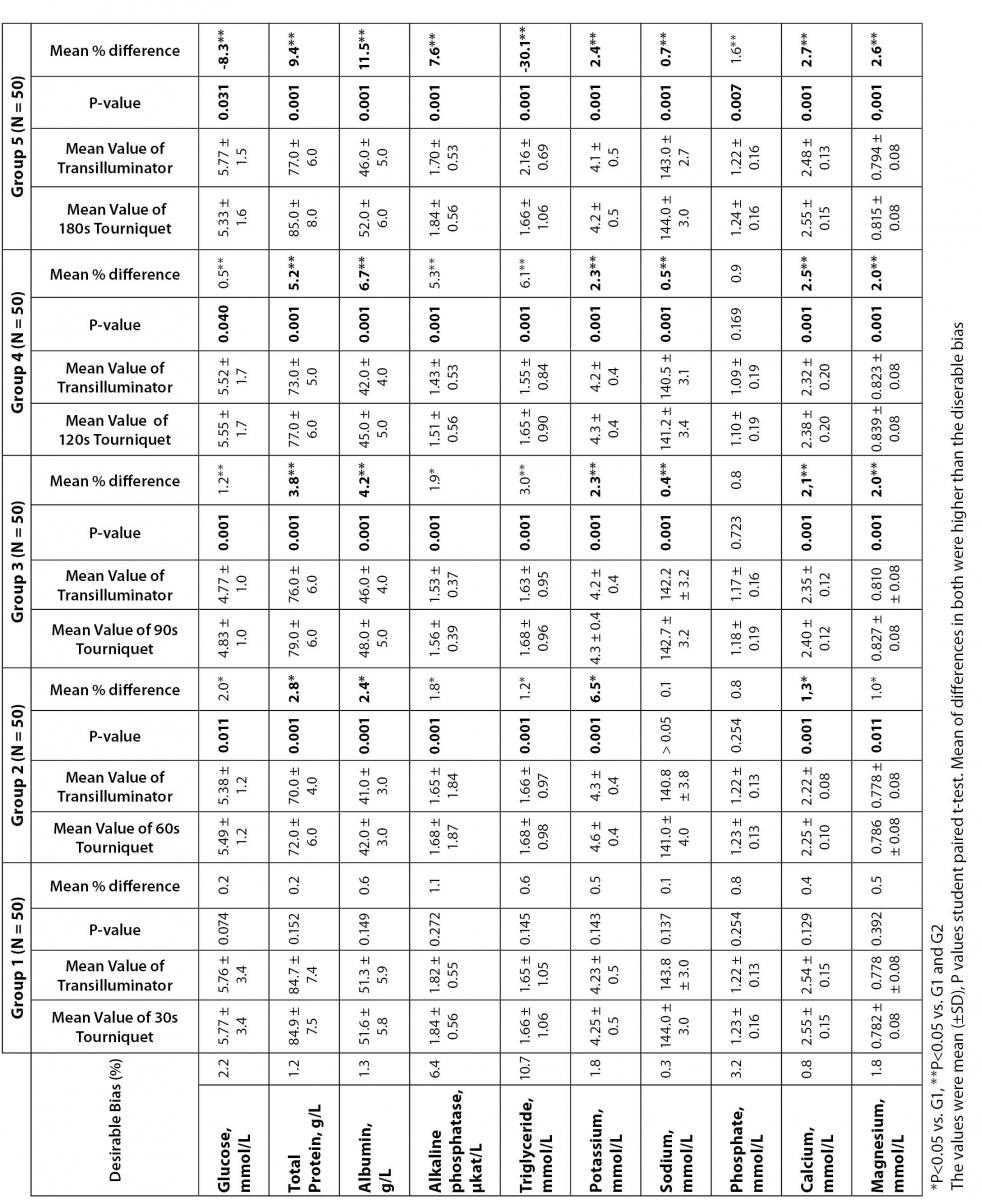
13. Lima-Oliveira GS, Picheth G, Assan NK, Ferreira CES, Mangueira CLP, Sumita NM, et al. The effects of tourniquet application vs. subctaneous tissue transilluminator device in blood sample collection on hematological parameters. Int J Lab Hematol 2007;29:37.
14. Lima-Oliveira GS, Picheth G, Assan NK, Ferreira CES, Sumita NM, Scartezini M. The effects of tourniquet application during 1 minute versus subcutaneous tissue transilluminator device in blood sample collection on biochemical parameters. Clin Chem 2007;53:123.
15. Lippi G, Salvagno GL, Montagnana M, Brocco G, Guidi GC. Influence of short-term venous stasis on clinical chemistry testing. Clin Chem Lab Med 2005;43:869-75.
16. Lippi G, Salvagno GL, Montagnana M, Franchini M, Guidi GC. Venous stasis and routine hematologic testing. Clin Lab Haematol 2006;28:332-7.
17. Lippi G, Salvagno GL, Montagnana M, Guidi GC. Short-term venous stasis influences routine coagulation testing. Blood Coagul Fibrinolysis 2005;16:453-8.
18. Lima-Oliveira GS, Picheth G, Sumita NM, Scartezini M. Phlebotomists performance during blood sample collection in public and private clinical laboratories in São Paulo city, Brazil. Clin Chem 2007;53:205.
19. Kuhns LR, Martin AJ, Gildersleeve S, Poznanski AK. Intense transillumination for infant venipuncture. Radiology 1975;116:734-5.
20. Goren A, Laufer J, Yativ N, Kuint J, Ben Ackon M, Rubinshtein M, et al. Transillumination of the palm for venipuncture in infants. Pediatr Emerg Care 2001;17:130-1.
21. Weiss RA, Goldman MP. Transillumination mapping prior to ambulatory phlebectomy. Dermatol Surg 1998;24:447-50.
22. Guder WG, Narayanan S, Wisser H, Zawta B. Diagnostic samples: from the patient to the laboratory: the impact of preanalytical variables on the quality of laboratory results. 4 ed: Wiley-Blackwell; 2009.
24. Ricos C, Alvarez V, Cava F, Garcia-Lario JV, Hernandez A, Jimenez CV, et al. Current databases on biological variation: pros, cons and progress. Scand J Clin Lab Invest 1999;59:491-500.
25. Lippi G, Lima-Oliveira G, Salvagno GL, Montagnana M, Gelati M, Picheth G, et al. Influence of a light meal on routine haematological tests. Blood Transfus 2010;8:94-9.
26. Loh TP, Saw S, Chai V, Sethi SK. Impact of phlebotomy decision support application on sample collection errors and laboratory efficiency. Clin Chim Acta 2011;412:393-5.
27. Gosselin RC, Janatpour K, Larkin EC, Lee YP, Owings JT. Comparison of samples obtained from 3.2% sodium citrate glass and two 3.2% sodium citrate plastic blood collection tubes used in coagulation testing. Am J Clin Pathol 2004;122:843-8.
28. Kratz A, Stanganelli N, Van Cott EM. A comparison of glass and plastic blood collection tubes for routine and specialized coagulation assays: a comprehensive study. Arch Pathol Lab Med 2006;130:39-44.
29. Lippi G, Salvagno GL, Montagnana M, Guidi GC. Influence of two different buffered sodium citrate concentrations on coagulation testing. Blood Coagul Fibrinolysis 2005;16:381-3.
30. Salvagno GL, Lippi G, Bassi A, Poli G, Guidi GC. Prevalence and type of pre-analytical problems for inpatients samples in coagulation laboratory. J Eval Clin Pract 2008;14:351-3.
31. van Geest-Daalderop JH, Mulder AB, Boonman-de Winter LJ, Hoekstra MM, van den Besselaar AM. Preanalytical variables and off-site blood collection: influences on the results of the prothrombin time/international normalized ratio test and implications for monitoring of oral anticoagulant therapy. Clin Chem 2005;51:561-8.
32. Polack B, Schved JF, Boneu B. Preanalytical recommendations of the ‘Groupe d’Etude sur l’Hemostase et la Thrombose’ (GEHT) for venous blood testing in hemostasis laboratories. Haemostasis 2001;31:61-8.
33. Salvagno GL, Lippi G, Montagnana M, Franchini M, Poli G, Guidi GC. Influence of temperature and time before centrifugation of specimens for routine coagulation testing. Int J Lab Hematol 2009;31:462-7.
34. Gren B. Incorrect guidelines for venipuncture affect the analytical results. Scand J Clin Lab Invest 2009;69:815-6.
35. Young DS. Effects of preanalytical variables on clinical laboratory tests 3ed. Washington: AACC Press; 2007.
36. Hallworth M, Hyde K, Cumming A, Peake I. The future for clinical scientists in laboratory medicine. Clin Lab Haematol 2002;24:197-204.
37. Dennis SC, Gevers W, Opie LH. Protons in ischemia: where do they come from; where do they go to? J Mol Cell Cardiol 1991;23:1077-86.
38. Lima-Oliveira G, Lippi G, Salvagno GL, Montagnana M, Scartezini M, Guidi GC, et al. Transillumination: a new tool to eliminate the impact of venous stasis during the procedure for the collection of diagnostic blood specimens for routine haematological testing. Int J Lab Hematol 2011 Mar 17 ŠEpub ahead of printĆ.
39. Lima-Oliveira G, Salvagno GL, Lippi G, Montagnana M, Scartezini M, Picheth G, et al. Elimination of the venous stasis error for routine coagulation testing by transillumination. Clin Chim Acta 2011 Apr 13 ŠEpub ahead of printĆ.
40. Lei B, Matsuo K, Labinskyy V, Sharma N, Chandler MP, Ahn A, et al. Exogenous nitric oxide reduces glucose transporters translocation and lactate production in ischemic myocardium in vivo. Proc Natl Acad Sci U S A 2005;102:6966-71.
41. Raymond RM, Harkema JM, Emerson TE, Jr. Mechanism of increased glucose uptake by skeletal muscle during E coli endotoxin shock in the dog. Circ Shock 1981;8:77-93.
42. Chandler MP, Huang H, McElfresh TA, Stanley WC. Increased nonoxidative glycolysis despite continued fatty acid uptake during demand-induced myocardial ischemia. Am J Physiol Heart Circ Physiol 2002;282:H1871-8.
43. Marcovina SM, Gaur VP, Albers JJ. Biological variability of cholesterol, triglyceride, low- and high-density lipoprotein cholesterol, lipoprotein(a), and apolipoproteins A-I and B. Clin Chem 1994;40:574-8.
44. Folmes CD, Sowah D, Clanachan AS, Lopaschuk GD. High rates of residual fatty acid oxidation during mild ischemia decrease cardiac work and efficiency. J Mol Cell Cardiol 2009;47:142-8.
45. Correia LC, Magalhaes LP, Braga JC, Rocha MS, Lima JC, Passos LC, et al. Decrease of plasma triglycerides during the acute phase of unstable angina or non-ST elevation myocardial infarction is a marker of recurrent ischemia. Atherosclerosis 2004;177:71-6