References
1. Rittirsch D, Flierl MA, Ward PA. Harmful molecular mechanisms in sepsis. Nature Rev Immunol 2008;8:776-87.
2. Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med 2003;31:1250-6.
3. Calandra T, Cohen J, International Sepsis Forum Definition of Infection in the ICUCC. The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Crit Care Med 2005;33:1538-48.
4. Dombrovskiy VY, Martin AA, Sunderram J, Paz HL. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: a trend analysis from 1993 to 2003. Crit Care Med 2007;35:1244-50.
5. Engel C, Brunkhorst FM, Bone HG, Brunkhorst R, Gerlach H, Grond S, et al. Epidemiology of sepsis in Germany: results from a national prospective multicenter study. Intens Care Med 2007;33:606-18.
6. Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 2006;34:1589-96.
7. Chan T, Gu F. Early diagnosis of sepsis using serum biomarkers. Expert Rev Mol Diagn 2011;11:487-96.
8. Faix JD. Established and novel biomarkers of sepsis. Biomarkers Med 2011;5:117-30.
9. Schiff DE, Rae J, Martin TR, Davis BH, Curnutte JT. Increased phagocyte Fc gammaRI expression and improved Fc gamma- receptor-mediated phagocytosis after in vivo recombinant human interferon-gamma treatment of normal human subjects. Blood 1997;90:3187-94.
10. Barth E, Fischer G, Schneider EM, Wollmeyer J, Georgieff M, Weiss M. Differences in the expression of CD64 and mCD14 on polymorphonuclear cells and on monocytes in patients with septic shock. Cytokine 2001;14:299-302.
11. Davis BH. Improved diagnostic approaches to infection/sepsis detection. Expert Rev Mol Diagn 2005;5:193-207.
12. Song SH, Kim HK, Park MH, Cho HI. Neutrophil CD64 expression is associated with severity and prognosis of disseminated intravascular coagulation. Thromb Res 2008;121:499-507.
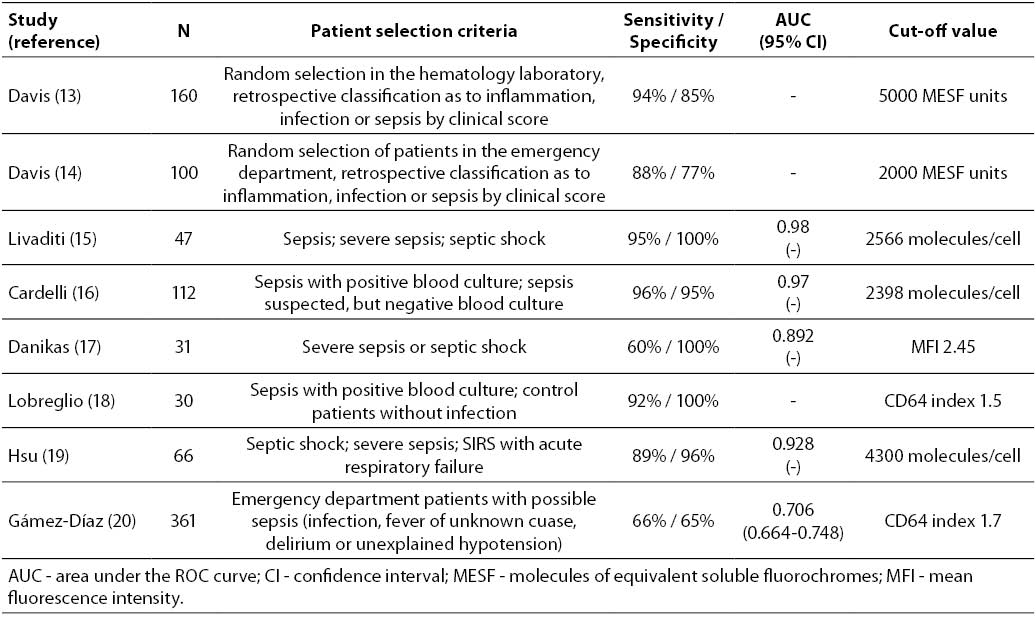
13. Davis BH, Bigelow NC. Comparison of neutrophil CD64 expression, manual myeloid immaturity counts, and automated hematology analyzer flags as indicators of infection or sepsis. Lab Hematol 2005;11:137-47.
14. Davis BH, Olsen SH, Ahmad E, Bigelow NC. Neutrophil CD64 is an improved indicator of infection or sepsis in emergencydepartment patients. Arch Pathol Lab Med 2006;130:654-61.
15. Livaditi O, Kotanidou A, Psarra A, Dimopoulou I, Sotiropoulou C, Augustatou K, et al. Neutrophil CD64 expression and serum IL-8: sensitive early markers of severity and outcome in sepsis. Cytokine 2006;36:283-90.
16. Cardelli P, Ferraironi M, Amodeo R, Tabacco F, de Blasi RA, Nicoletti M, et al. Evaluation of neutrophil CD64 expression and procalcitonin as useful markers in early diagnosis of sepsis. Int J Immunopathol Pharmacol 2008;21:43-9.
17. Danikas DD, Karakantza M, Theodorou GL, Sakellaropoulos GC, Gogos CA. Prognostic value of phagocytic activity of neutrophils and monocytes in sepsis Correlation to CD64 and CD14 antigen expression. Clin Exp Immunol 2008;154:87-97.
18. Lobreglio GB, d’Aversa P, Leo L, Scolozzi S, Fiore G. Quantitative expression of CD64 on neutrophil granulocytes as early marker of sepsis or severe infection. Haematologica 2008;93:21.
19. Hsu KH, Chan MC, Wang JM, Lin LY, Wu CL. Comparison of Fcgamma receptor expression on neutrophils with procalcitonin for the diagnosis of sepsis in critically ill patients. Respirol 2011;16:152-60.
20. Gámez-Díaz LY, Enriquez LE, Matute JD, Velásquez S, Gómez ID, Toro F, et al. Diagnostic Accuracy of HMGB-1, sTREM-1, and CD64 as Markers of Sepsis in Patients Recently Admitted to the Emergency Department. Acad Emerg Med 2011;18:807-15.
21. Hoffmann JJML. Neutrophil CD64: a diagnostic marker for infection and sepsis. Clin Chem Lab Med 2009;47:903-16.
22. Cid J, Aguinaco R, Sánchez R, García-Pardo G, Llorente A. Neutrophil CD64 expression as marker of bacterial infection: a systematic review and meta-analysis. J Inf 2010;60:313-9.
23. Ng PC. Diagnostic markers of infection in neonates. Archives of Disease in Childhood 2004;89:F229-35.
24. Arnon S, Litmanovitz I. Diagnostic tests in neonatal sepsis. Curr Opin Infect Dis 2008;21:223-7.
25. Fjaertoft G, Håkansson L, Foucard T, Ewald U, Venge P. CD64 Fcgamma receptor I cell surface expression on maturing neutrophils from preterm and term newborn infants. Acta Paediatr 2005;94:295-302.
26. Nuutila J. The novel applications of the quantitative analysis of neutrophil cell surface FcgammaRI CD64 to the diagnosis of infectious and inflammatory diseases. Curr Opin Infect Dis 2010;23:268-74.
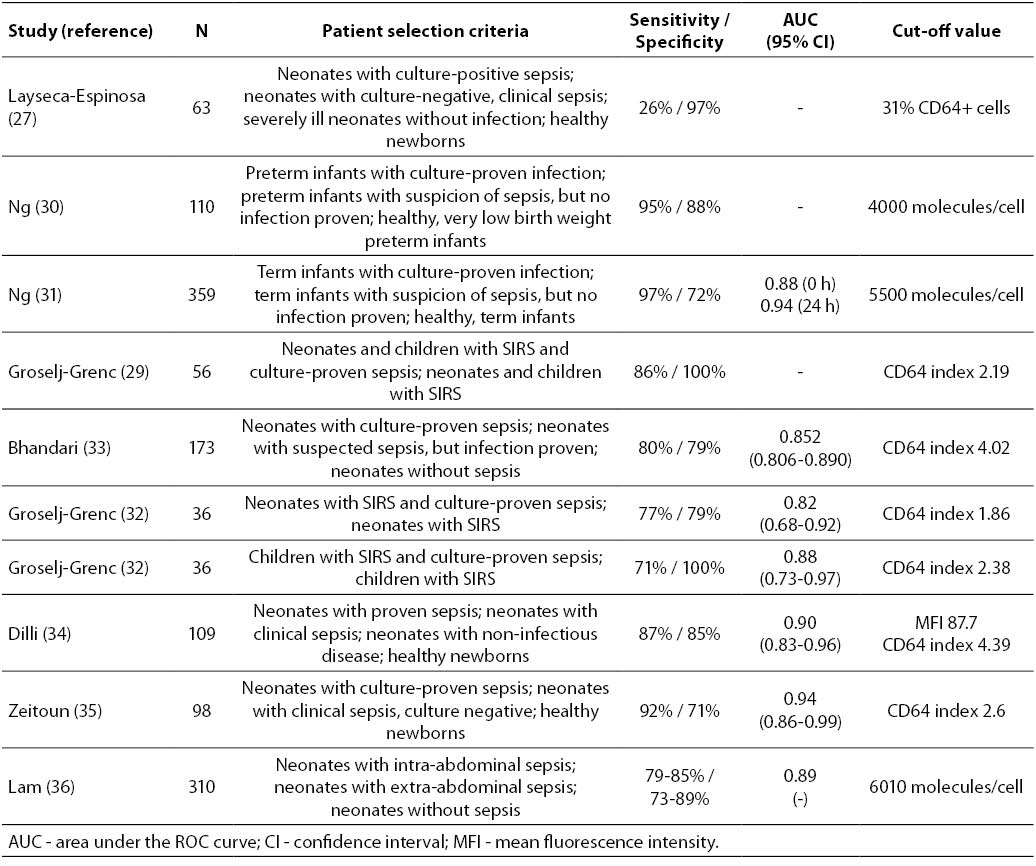
27. Layseca-Espinosa E, Pérez-González LF, Torres-Montes A, Baranda L, de la Fuente H, Rosenstein Y, González-Amaro R. Expression of CD64 as a potential marker of neonatal sepsis. Ped Allergy Immunol 2002;13:319-27.
28. Fjaertoft G, Håkansson LD, Pauksens K, Sisask G, Venge P. Neutrophil CD64 FcgammaRI expression is a specific marker of bacterial infection: a study on the kinetics and the impact of major surgery. Scand J Inf Dis 2007;39:525-35.
29. Groselj-Grenc M, Ihan A, Derganc M. Neutrophil and monocyte CD64 and CD163 expression in critically ill neonates and children with sepsis: comparison of fluorescence intensities and calculated indexes. Mediat Inflamm 2008;2008:202646.
30. Ng PC, Li K, Wong RPO, Chui KM, Wong E, Fok TF. Neutrophil CD64 expression: a sensitive diagnostic marker for late- onset nosocomial infection in very low birthweight infants. Ped Res 2002;51:296-303.
31. Ng PC, Li G, Chui KM, Chu WCW, Li K, Wong RPO, et al. Neutrophil CD64 is a sensitive diagnostic marker for early-onset neonatal infection. Ped Res 2004;56:796-803.
32. Groselj-Grenc M, Ihan A, Pavcnik AM, Kopitar AN, Gmeiner ST, Derganc M. Neutrophil and monocyte CD64 indexes, lipopolysaccharide-binding protein, procalcitonin and C-reactive protein in sepsis of critically ill neonates and children. Intens Care Med 2009;35:1950-8.
33. Bhandari V, Wang C, Rinder C, Rinder H. Hematologic profile of sepsis in neonates: neutrophil CD64 as a diagnostic marker. Pediatrics 2008;121:129-34.
34. Dilli D, Oguz ŞS, Dilmen Uu, Köker MY, Kızılgün M. Predictive values of neutrophil CD64 expression compared with interleukin-6 and C-reactive protein in early diagnosis of neonatal sepsis. J Clin Lab Anal 2010;24:363-70.
35. Zeitoun AAH, Gad SS, Attia FM, Abu Maziad AS, Bell EF. Evaluation of neutrophilic CD64, interleukin 10 and procalcitonin as diagnostic markers of early- and late-onset neonatal sepsis. Scand J Inf Dis 2010;42:299-305.
36. Lam HS, Wong SPS, Cheung HM, Chu WCW, Wong RPO, Chui KM, et al. Early diagnosis of intra-abdominal inflammation and sepsis by neutrophil CD64 expression in newborns. Neonatol 2011;99:118-24.
37. Icardi M, Erickson Y, Kilborn S, Stewart B, Grief B, Scharnweber G. CD64 index provides simple and predictive testing for detection and monitoring of sepsis and bacterial infection in hospital patients. J Clin Microbiol 2009;47:3914-9.
38. Tanaka S, Nishino J, Matsui T, Komiya A, Nishimura K, Tohma S. Neutrophil CD64 expression in the diagnosis of local musculoskeletal infection and the impact of antibiotics. J Bone Joint Surg 2009;91:1237-42.
39. Fischer G, Schneider EM, Moldawer LL, Karcher C, Barth E, Suger-Wiedeck H, et al. CD64 surface expression on neutrophils is transiently upregulated in patients with septic
shock. Intens Care Med 2001;27:1848-52.
40. Muller Kobold AC, Tulleken JE, Zijlstra JG, Sluiter W,
Hermans J, Kallenberg CG, Tervaert JW. Leukocyte activation in sepsis; correlations with disease state and mortality.
Intens Care Med 2000;26:883-92.
41. Cid J, García-Pardo G, Aguinaco R, Sánchez R, Llorente A. Neutrophil CD64: diagnostic accuracy and prognostic value in patients presenting to the emergency department. Eur J Clin Microbiol Inf Dis 2011;30:845-52.
42. Allen E, Bakke AC, Purtzer MZ, Deodhar A. Neutrophil CD64 expression: distinguishing acute inflammatory autoimmune disease from systemic infections. Ann Rheum Dis 2002;61:522-5.
43. Doi T, Miyazaki T, Nishino J, Tanaka S, Matsui T, Komiya A, et al. Neutrophil CD64 expression as a diagnostic marker for local infection and crystal-induced arthritis. Modern Rheumatol 2010;20:573 -9.
44. Hussein OA, El Toukhy MA, El Rahman HS. Neutrophil CD64 expression in inflammatory autoimmune diseases: its value in distinguishing infection from disease flare. Immunol
Invest 2010;39:699-712.
45. Jalava-Karvinen P, Hohenthal U, Laitinen I, Kotilainen P, Rajamäki A, Nikoskelainen J, et al. Simultaneous quantitative analysis of FcŠgammaĆRI (CD64) and CR1 (CD35) on neutrophils in distinguishing between bacterial infections, viral infections, and inflammatory diseases. Clin Immunol 2009;133:314-23.
46. Matsui T, Ohsumi K, Ozawa N, Shimada K, Sumitomo S, Shimane K, et al. CD64 on neutrophils is a sensitive and specific marker for detection of infection in patients with rheumatoid arthritis. J Rheumatol 2006;33:2416-24.
47. Nishino J, Tanaka S, Kadono Y, Matsui T, Komiya A, Nishimura K, Tohma S. The usefulness of neutrophil CD64 expression in the diagnosis of local infection in patients with rheumatoid arthritis in daily practice. J Orthoped Sci 2010;15:547-52.
48. Strohmeyer JC, Blume C, Meisel C, Doecke WD, Hummel M, Hoeflich C, et al. Standardized immune monitoring for the prediction of infections after cardiopulmonary bypass surgery in risk patients. Cytometry 2003;53B:54-62.
49. Daryapeyma A, Pedersen G, Laxdal E, Corbascio M, Johannessen HB, Aune S, Jonung T. Neutrophil CD64 as a marker for postoperative infection: a pilot study. Eur J Vasc Endovasc Surg 2009;38:100-3.
50. Tillinger W, Jilch R, Jilma B, Brunner H, Koeller U, Lichtenberger C, et al. Expression of the high-affinity IgG receptor FcRI CD64 in patients with inflammatory bowel disease: a new biomarker for gastroenterologic diagnostics. Am J Gastroenterol 2009;104:102-9.
51. Koga T, Umeda M, Migita K, Yamasaki S, Nakamura H, Kawakami A. A Japanese case of familial Mediterranean fever presenting diffuse bone marrow uptake of FDG-PET and high levels of neutrophil membrane CD64 expression. Rheumatol 2011;50:1171-3.
52. Kamprad M, Grey D, Bauer K, Knaack H, Fangmann J, Emmrich F, Sack U. Detection of complications following solid organ transplantation by CD64 on PMNs. Cytometry 2008;74B:411-2.
53. Bakke AC, Allen E, Purtzer MZ, Deodhar A. Neutrophil CD64 expression distinguishing acute inflammatory autoimmune disease from systemic infections. Clin Appl Immunol Rev 2001;1:267-75.
54. Davis B, Becker K, Rinder H, Davis K. Neutrophil CD64 expression, a marker of sepsis/infection, can be performed on a hematology blood counter and has variable correlation to C-reactive protein, procalcitonin and soluble CD163. Crit Care 2007;11(suppl 4):S9-S10.
55. Grey D, Sack U, Scholz M, Knaack H, Fricke S, Oppel C, et al. Increased CD64 expression on polymorphonuclear neutrophils indicates infectious complications following solid
organ transplantation. Cytometry 2011;79A:446-60.
56. Geaghan S, Nava A, George T. CD64 quantitation on neutrophils using a conventional hematology analyzer for adult and pediatric specimens in a tertiary care hospital and clinics setting. Int J Lab Hematol 2011;33 (Suppl. 1):60.
57. Pavic M, Bronic A, Milevoj Kopcinovic L. Procalcitonin in systemic and localized bacterial infection. Biochem Med 2010;20:236-41.